42 lot number on medication label
CFR - Code of Federal Regulations Title 21 - Food and Drug Administration The lot number on the label of a drug should be capable of yielding the complete manufacturing history of the package. An incorrect lot number may be regarded as causing the article to be misbranded. Sec. 201.19 Drugs; use of term "infant". The regulations affecting special dietary foods (§ 105.3(e) of this chapter) define an infant as a child ... How to Read Medication Orders and Drug Labels - Oregon.gov Labels on prescription medications;. • Over-the-counter (OTC) medications; ... A lot has changed in the last 10 years. ... Number of refills allowed;.
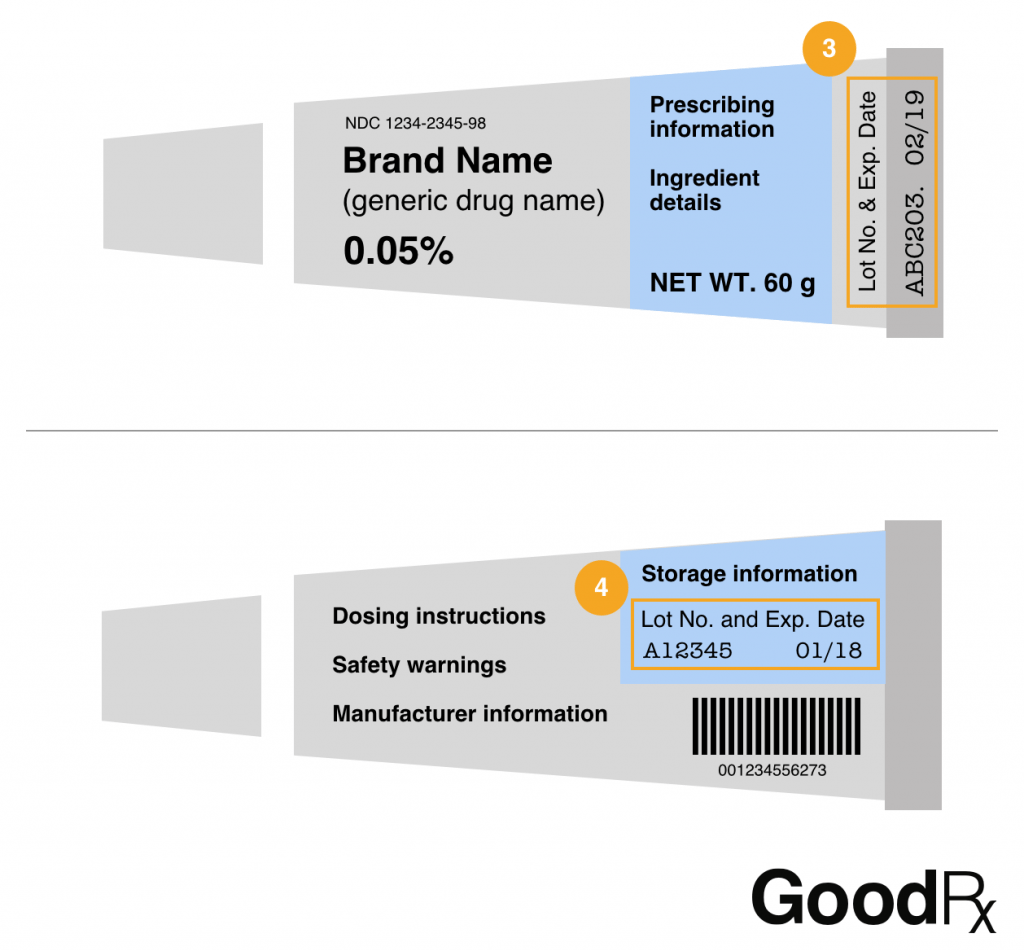
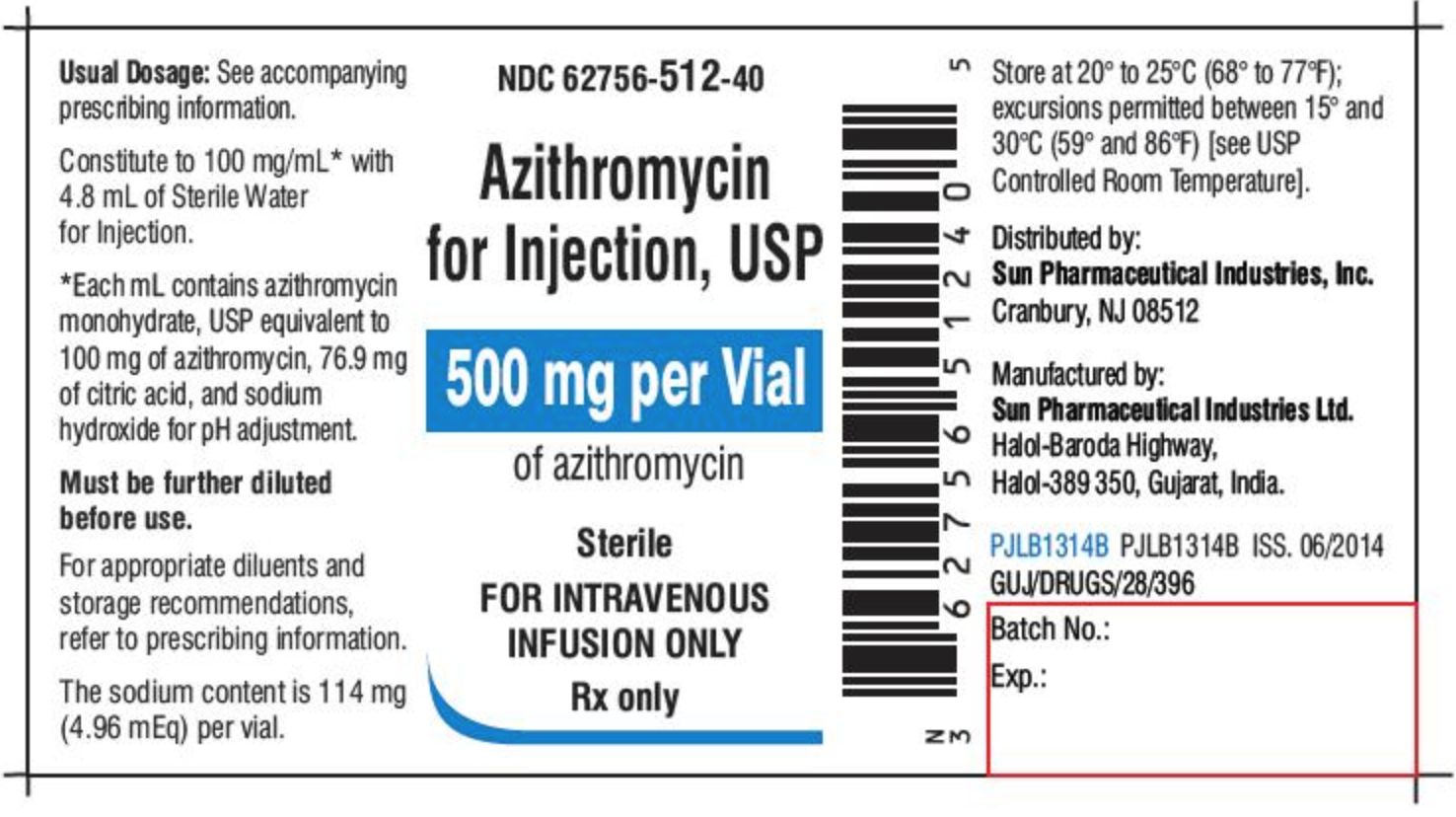
Drug Recalls: What to Do and How to Find Your Medication ... Feb 12, 2019 ... Drug bottles will often have the lot number printed next to the expiration date, either by the barcode (1) or underneath the dosing instructions ...

Lot number on medication label
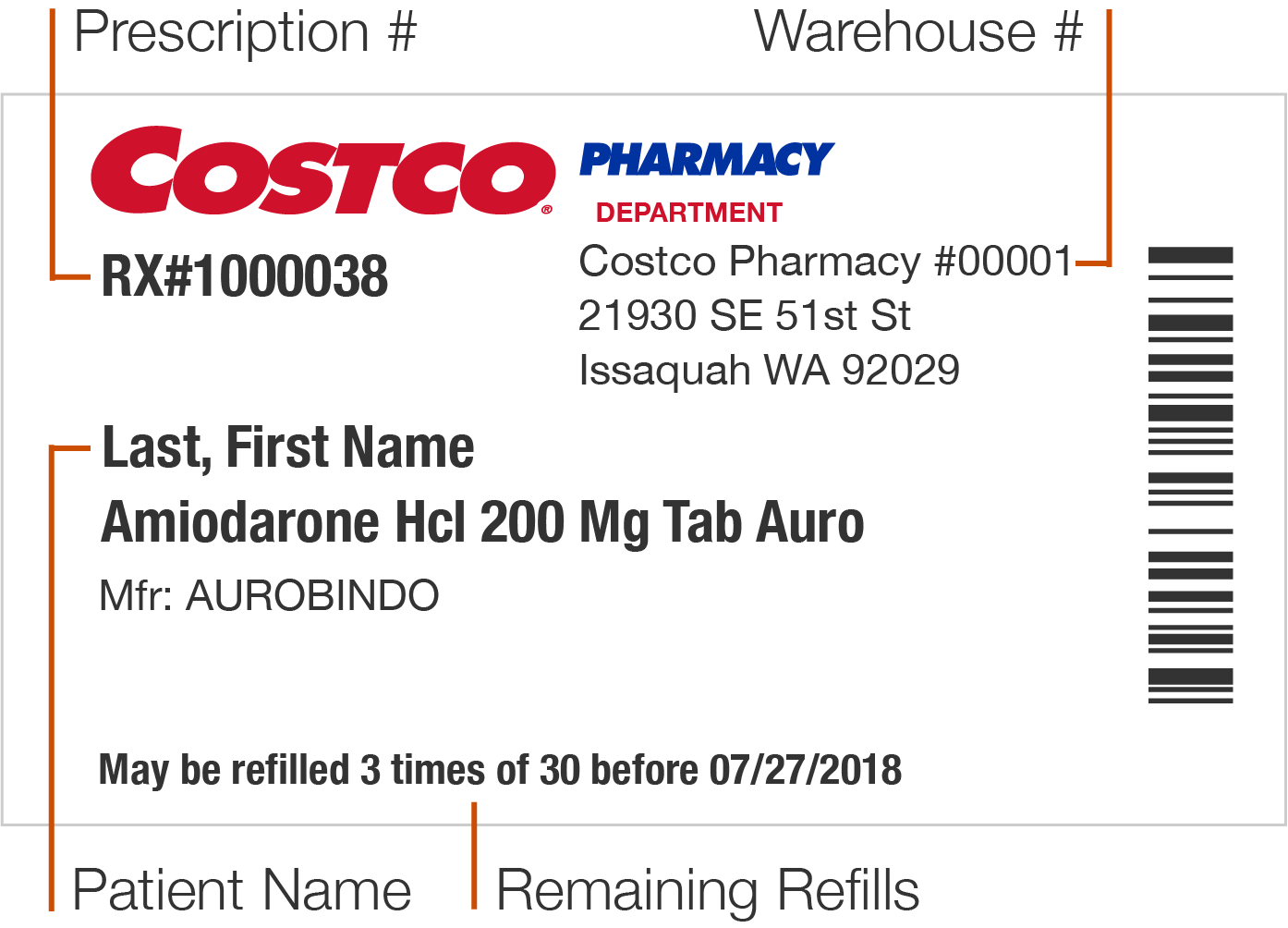
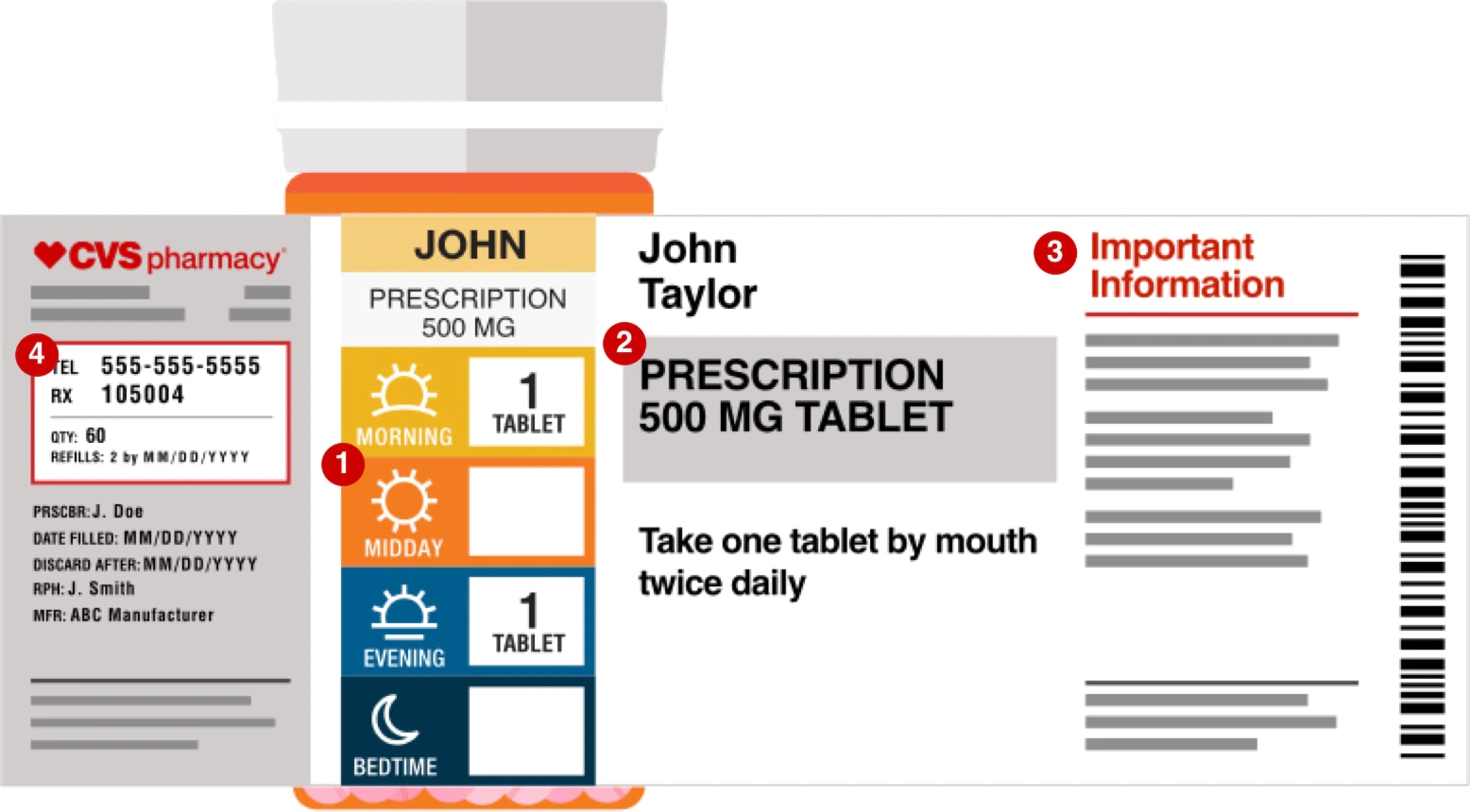
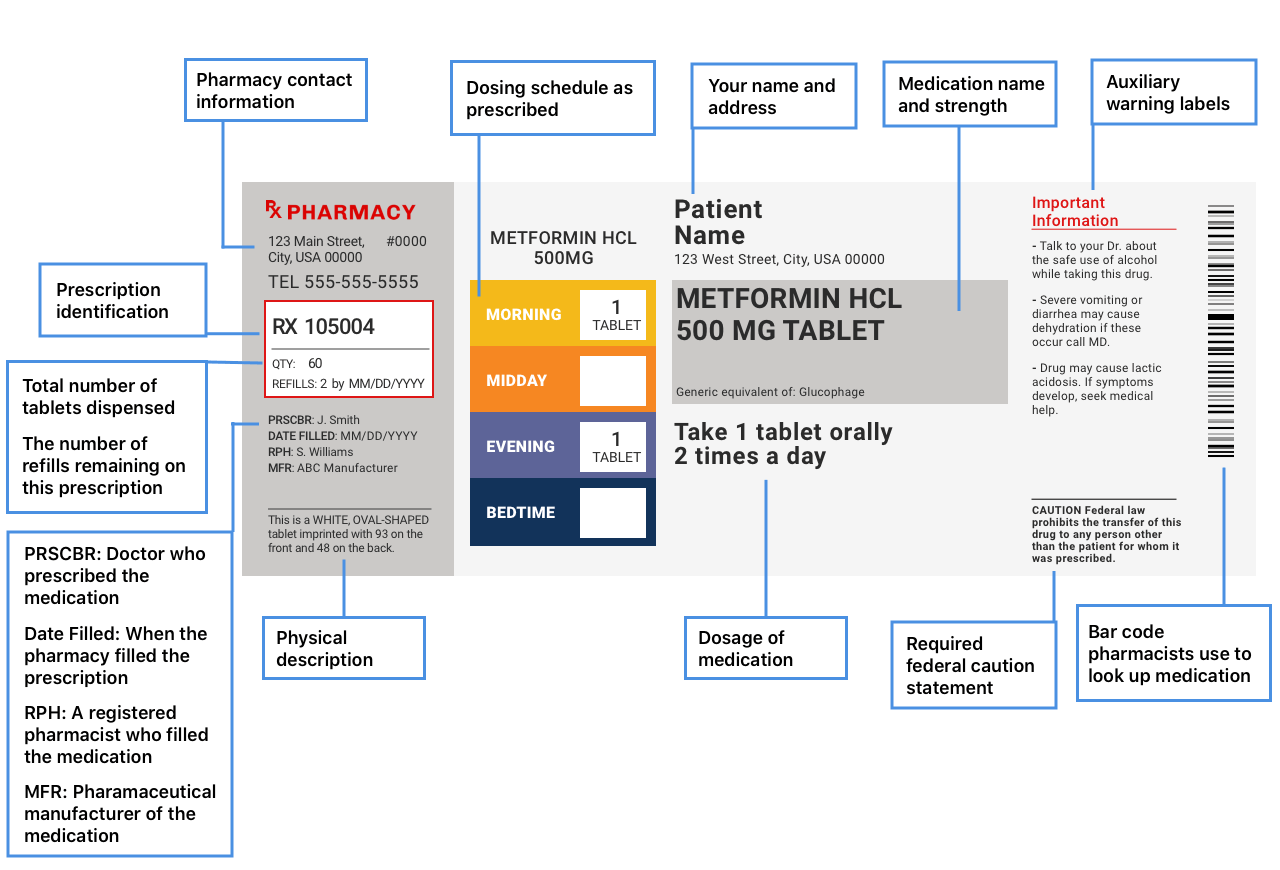
Prescription labels and how to read them Your pharmacy name, address, and phone number · Your prescription number (RX#) · Your name · The date your prescription was filled · The name of the prescriber · How ... Is NDC code required to appear on both 'bottle carton' and 'label' of ... As per the CFR regulation 21 Title CFR 201.2, "NDC number is requested but not required to appear on all drug labels and in all drug labeling, including the label of any prescription drug container furnished to a consumer." but our concern is specifically for the consistency of presence of NDC code on outer and inner labels for a bottle. Prescription Label Information, Translations, and Sample Labels ... Prescription Label Information, Translations, and Sample Labels. Translations of Pill Directions ; Patient-Centered Prescription Drug Container Label Samples; Prescription Drugs: Labeling Requirements - Report to the Legislature; Statutory Requirements (4076.5) and Regulation Requirements (1707.5)
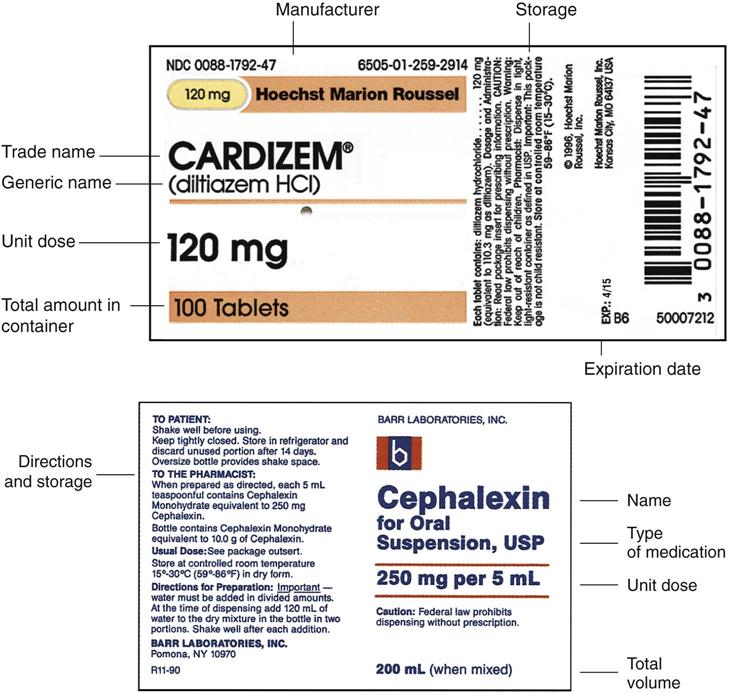
Lot number on medication label. Lot number | definition of lot number by Medical dictionary lot number An identifier assigned to a batch of medications. It facilitates drug manufacturing inventory control and tracing adverse incidents in a batch of contaminated medications. See also: number Medical Dictionary, © 2009 Farlex and Partners Want to thank TFD for its existence? PDF Attachment 15: Medication Container Labeling - mecknc.gov the filling of the medication) • Rx number and date filled • Name of prescriber • Directions for use • Name (brand and generic) of medication ... This can be done by using color ink on the labels or attaching a colored sticker to the bottle. Patient information could then be printed on ... Lot # SAMPLE EXP: 00-00 Rx 5220953 ... Label mix up prompts recall of blood pressure and heart drugs After receiving a report that the label was wrong on a medication, Golden State Medical Supply has recalled one lot of clopidogrel and atenolol. According to an announcement with the U.S. Food and... Medication Errors in a Pharmacy-Based Bar-Code-Repackaging - Medscape NDC b number, lot number, expiration date, drug name, strength, dosage form, ... Incomplete label: 100 (1.3) 560 (7.4) 660 (8.8) Incorrect medication: 0: 6 (0.1) 6 (0.1) Total errors detected:
Lot Pill Images - Pill Identifier - Drugs.com All prescription and over-the-counter (OTC) drugs in the U.S. are required by the FDA to have an imprint code. If your pill has no imprint it could be a vitamin, diet, herbal, or energy pill, or an illicit or foreign drug. It is not possible to accurately identify a pill online without an imprint code. Learn more What Information Should Be on Drug Labels? - MedicineNet Before any drug can be legally sold, it must be assigned a Drug Identification Number (DIN). The FDA recommends manufacturers to provide data demonstrating product safety, effectiveness in meeting label claims, potency, and purity. When the manufacturer meets these requirements, the FDA assigns the drug a registration number. The Importance of Lot Tracking for Pharmaceutical Manufacturers What is Lot Tracking? · which batches of medications are ready for purchase · which batches expired and, if necessary, · which batches of medication need to be ... FDA Label Search - Food and Drug Administration The drug labels and other drug-specific information on this Web site represent the most recent drug listing information companies have submitted to the Food and Drug Administration (FDA). (See 21 CFR part 207.) The drug labeling and other information has been reformatted to make it easier to read but its content has neither been altered nor ...
How to read prescription drug labels - BeMedwise All prescription medicine containers include information on the label including the patient's name, the name of the medicine, dosage and instructions on how ... How to Read Over the Counter Drug Labels - Verywell Health The U.S. Food and Drug Administration (FDA) requires all over-the-counter (OTC) medications to have a Drug Facts label. This label provides basic information about a drug's ingredients, instructions for use, and important safety cautions and interactions. This information will help you to select the correct medication and to use it properly. 1. The Over-the-Counter Medicine Label: Take a Look | FDA The Label Also Tells You... The expiration date, when applicable (date after which you should not use the product). Lot or batch code (manufacturer information to help identify the product). Name... What is a lot number on medication? - Quora Lot numbers are not assigned by the FDA. They are numbers assigned by a manufacturer to identify a batch of a particular medication that was produced. The lot ...
Understand your medication label | Express Scripts® Pharmacy Nov 30, 2021 ... We've broken down the important parts of a prescription label to help you ... Call this number for any questions about your medication.
CVS pharmacy - Frequently Asked Questions Your label has the following information: Your name Your prescription name and number Directions for use The quantity of the medication The number of refills and prescription expiration date The phone number and store number of the CVS/pharmacy where you filled the prescription Your prescriber's name
How to Read a Medication (Drug) Label for Nurses These two numbers are typically located together. The lot number is helpful with identifying recalled medications and to help the manufacturer track the ...
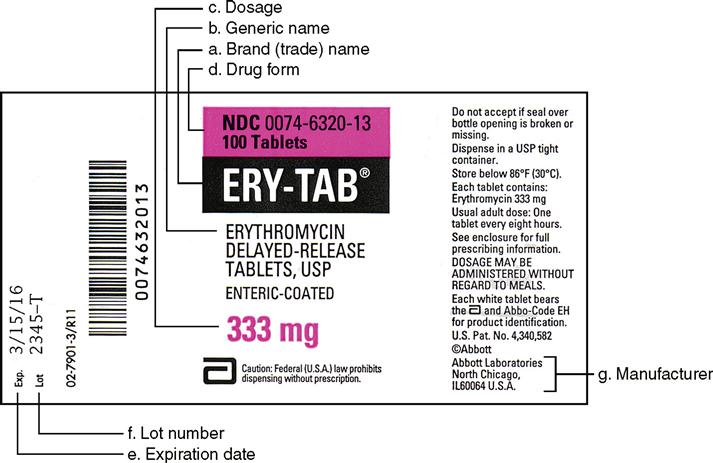
PDF Chapter 20 Labeling Medications and Expiration Dating A. UNIT DOSE MEDICATION - (Prepackaging) reference 64F-12.006 Minimum labeling to include: a) Name of drug (brand or generic or both) b) Strength c) Dosage Form d) Manufacturer e) Lot number f) Expiration date/beyond use date g) OR instead of (d) and (e) a control number which cross references to the manufacturer name and lot number
Prescription Labels and Drug Safety - Consumer Reports Prescription bottle labels. There are about 1.5 million preventable medication errors each year, according to a 2006 report from the Institute of Medicine. Roughly one-third of those mistakes ...
21 CFR § 203.38 - Sample lot or control numbers; labeling of sample ... The manufacturer or authorized distributor of record of a drug sample shall include on the label of the sample unit and on the outside container or packaging of the sample unit, if any, an identifying lot or control number that will permit the tracking of the distribution of each drug sample unit.
What's on my medicine label? - Therapeutic Goods Administration (TGA) The label will include contact details for either the medicine's sponsor or distributor, including the company name and suburb. There could also be a phone number or website address. This information can be used to make an enquiry or complaint, or report a side effect. Australian Register of Therapeutic Goods (ARTG) number
NCBOP - Pharmacist FAQs Q. What information is required to be on the prescription label? A. The following information must be on every prescription label: 1. Name and address of the dispensing pharmacy. 2. Serial number of the prescription. 3. Date of the prescription. 4. Name of the prescriber. 5. Name of the patient. 6. Name and strength of the drug. 7.
How to Understand FDA Lot Numbers | Healthfully The lot number may or may not be labeled as such; in other words, you may see "lot" followed by the number, but not always. When looking for the lot number, keep in mind that it will not be printed as a standard part of the product label since the product label doesn't change, whereas the lot number changes with every batch.
What is a lot number on medication? - Quora It's a multi digit or letter and digit number on a label , usually near the expiration date. Many medications require this in the event a bad lot (batch) turns up later. It's a quality control number, indicating that one or more bottles in that batch have been tested and the batch is okay for distribution. Sponsored by Phytage Labs
Quality System Regulation Labeling Requirements | FDA Most labeling, however, also contains another number, such as a drawing number, for control of labeling configuration and procurement. The control number for traceability need not be on every label...
Amazon.com: Medication Labels - Recordkeeping & Labels: Industrial ... Narcotics Medication Label Tape FENTANYL_mcg/mL 2 $2100 ($0.04/Count) FREE delivery Oct 6 - 12 PHARMEX 1-369 Permanent Paper Label,"Date Opened", 1 9/16" x 3/8", Yellow (500 per Roll, 2 Rolls per Box) 36 $1261 ($0.01/Count) FREE delivery Thu, Oct 6 on $25 of items shipped by Amazon Or fastest delivery Mon, Oct 3 Subscribe & Save $1261 ($0.01/Count)
How To Read A Prescription Bottle Label | Prescription Number - CVS The label should show your name prominently. Also confirm that the bottle lists the same medication and instructions that your provider explained when prescribing, and always take medication exactly as your provider prescribes. You can also consider Spoken Rx™ to have your prescription labels read out loud to you.
FDA Label Search-Package Code - Food and Drug Administration FDA Label Search FDA Home Search by NDC: (Type the 4 or 5 digit NDC Labeler Code with the hyphen (e.g., 0001-), the 8 or 9 digit NDC Product Code (e.g., 0001-0001) or the 10 digit NDC...
Understanding Drug Labels | Basicmedical Key Jun 24, 2016 ... Federal law requires that all medication packages be identified with a lot number or control number. Lot numbers are used to track products in ...
PDF Chapter 20 Labeling Medications and Expiration Dating (f) Lot number: 1. Manufacturer's lot number, or 2. Number assigned by the dispenser or repackager which references the manufacturer's lot number. (4) A medicinal drug dispensed in a unit dose system by a pharmacist shall be accompanied by labeling.
Federal Register :: Potential Medication Error Risks With ... While not a regulatory requirement, some investigational new drug container labels may include additional information such as the protocol/clinical trial number, concentration and/or strength, dosage form (e.g., tablets, injection), quantity per container, storage requirements, and lot number.
How To Read A Prescription Label | CenterWell Pharmacy™ Prescription labels pack a lot of information into a small space. Let's take a closer look at a ... If you refill, you'll need to provide your Rx number.
Guidance Document: Labelling of Pharmaceutical Drugs for Human Use 3.4.6 Drug Identification Number 3.5 Any Panel 3.5.1 Name and Address of Manufacturer/Sponsor 3.5.2 Lot Number 3.5.3 Expiration Date 3.5.4 Adequate Directions for Use 3.5.4.1 Storage Conditions 3.5.4.2 Limit Dose Drug Products 3.5.4.3 Warnings and Precautions 3.5.5 Declaration of Medicinal Ingredients
Drug labelling - Wikipedia In general, the expiry date of the drug is required and shall appear on the mediate container and the outer package. [5] Additional, label statements should be displayed with prominence and conspicuousness. The lot number, also called batch number, on the label should generate the full manufacturing history of the package. [5]
What is a lot number and how do I identify it? - Acme United ... The lot number identifies when a product was manufactured. It will be in small type, all capital letters, and say "LOT #". On many products, it's on the back panel. On kit boxes, it's either on the top or bottom of the box, generally affixed to a label. Lot numbers have 2 parts: a letter, followed by a series of numbers.
Expiry Dates on Prescription Labels - OCPInfo.com C.01.004. (1) The inner and outer labels of a drug shall show c) on any panel. the name and address of the manufacturer of the drug, the lot number of the drug, adequate directions for use of the drug, a quantitative list of the medicinal ingredients of the drug by their proper names or, if they have no proper names, by their common names,
Prescription Label Information, Translations, and Sample Labels ... Prescription Label Information, Translations, and Sample Labels. Translations of Pill Directions ; Patient-Centered Prescription Drug Container Label Samples; Prescription Drugs: Labeling Requirements - Report to the Legislature; Statutory Requirements (4076.5) and Regulation Requirements (1707.5)
Is NDC code required to appear on both 'bottle carton' and 'label' of ... As per the CFR regulation 21 Title CFR 201.2, "NDC number is requested but not required to appear on all drug labels and in all drug labeling, including the label of any prescription drug container furnished to a consumer." but our concern is specifically for the consistency of presence of NDC code on outer and inner labels for a bottle.
Prescription labels and how to read them Your pharmacy name, address, and phone number · Your prescription number (RX#) · Your name · The date your prescription was filled · The name of the prescriber · How ...



























Post a Comment for "42 lot number on medication label"